![[Image: masthead]](/mhau001/includes/themes/MHAUS_2016/images/masthead/masthead-large.png)
24-HOUR MH HOTLINE: 800-644-9737
Outside NA: 001-209-417-3722
FOR EMERGENCIES ONLY
Outside NA: 001-209-417-3722
FOR EMERGENCIES ONLY
Post Acute Phase
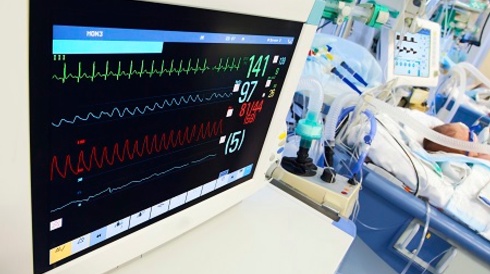
Post Acute Phase Management in PACU or ICU
- If assistance is needed, call the MHAUS Hotline (1-800-644-9737).
- Monitor ECG, ETCO2, minute ventilation, core temperature, urine output (with bladder catheter if necessary), and consider arterial and/or central venous monitoring if warranted by the clinical severity of the patient. Document muscle tone.
- Follow continuously for the first 24 hours:
- HR
- RR
- Core temperature
- ETCO2 (if intubated)
- Oxygen saturation
- Muscle tone
- Follow:Every 8 hours:
- Blood pH and lactate
- K+
- CK (until decreasing)
- Other:
- Coagulation studies should be obtained as a baseline and when there is evidence of bleeding
- Baseline renal function, i.e., creatinine - should be measured and followed at least q 24h if myoglobinuria has occurred
- A search for infectious causes for a hypermetabolic state may be appropriate
- Follow urine color and institute therapy to prevent myoglobin precipitation in renal tubules and acute renal failure. CK levels above 10,000 IU/L are a presumptive sign of rhabdomyolysis and myoglobinuria.
- Alkalinize urine with sodium bicarbonate, use diuretics to maintain urine output >2ml/kg/hr.
- Follow urine and serum pH values, serum electrolytes, and creatinine.
- Watch for MH relapse by continuously evaluating the patient for at least 24 hours following cessation of signs of MH. 25% of MH events relapse and relapses can be fatal. Treat immediately if relapse occurs. Signs of MH relapse include:
- Increasing muscular rigidity in the absence of shivering
- Inappropriate hypercarbia with respiratory acidosis
- Metabolic acidosis without other cause
- Inappropriate temperature rise
- Give dantrolene, 1mg/kg IV q 4-6 hours. Continue for at least 24 hours and sometimes longer as clinically indicated. Watch the IV site for evidence of thrombophlebitis or extravasation of the drug and attendant tissue necrosis. Manage the affected site according to your facility protocol. Continue dantrolene treatment using a new IV site. Dantrolene can be stopped, or the interval between doses increased to q8 or q12h if all of the following criteria are met:
- Metabolic stability for 24 hours
- Core temp is less than 38°C
- CK is decreasing
- No evidence of myoglobinuria
- Muscle is no longer rigid
- Monitor for MH Complications:
- Change in consciousness level/coma
- Cardiac dysrhythmias and dysfunction
- Pulmonary edema
- Renal dysfunction due to acute tubular necrosis
- Disseminated intravascular coagulation
- Hepatic dysfunction
- Muscular weakness (Secondary to both rhabdomyolysis and dantrolene)
- Compartment syndrome (Secondary to rhabdomyolysis)
Report this anesthetic complication on the AMRA form to the MH Registry of MHAUS. Printable PDF forms are available online via the registry area of our site, or by contacting:
The North American MH Registry of MHAUS
University of Florida
Department of Anesthesiology
Communicore Building
1345 SW Center Drive
Second Floor, Room C2-22
PO Box 100254
Gainesville, FL 32611-0254
1-888-274-7899
http://anest.ufl.edu/namhr/
http://anest.ufl.edu/namhr/
Alert the family to the dangers of MH in the other family members. Refer family members for testing at the nearest MH Diagnostic Testing Center. See our testing page for a list of MH Diagnostic Testing Centers. Individuals who experienced fulminant MH episodes should have blood sent for genetic screen of the entire RYR1.